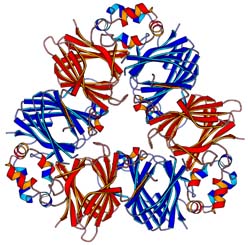
 The structure of canavalin from Jack Bean
The structure of canavalin from Jack Bean
Four crystal forms of this vicilin-class storage protein were determined in
collaboration with Prof. Alex McPherson by Multiple Isomorphous Replacement
and Molecular Replacement methods. The structure is a trimer of 142,000 Da
in total and each subunit has Mr = 47,000 Da. A subunit is divided into two
very similar, structurally homologous domains, each contains a core barrel
subdomain of Swiss Roll topology and a loop subdomain with three helices.
The barrels are packed back to back by a strong hydrophobic interface and the
monomers are associated via the other face of the barrels and the extended
loop subdomains. This protein is homologous to many other seed proteins,
including phaseolin from French bean whose crystal structure has also been
determined. The homology can be extended to another major class of storage
proteins - legumin. Recent characterization of a sucrose binding protein from
soy bean shows high sequence homology to canavalin. The exact biological
function of vicilin-class proteins as well as their evolution origin are yet
to be elucidated. Shown in red is the N-terminal domain and in blue is the
C-terminal domain. Coordinates are available from PDB entries 2CAU and 2CAV,
1DGR and 1DGW, and also 1CAU, 1CAV, 1CAW and 1CAX.
See:
Acta Cryst. D56, 411-420.
Copyright © International Union of Crystallography
Reprint in PDF format
Acta Cryst. D57, 829-839.
Copyright © International Union of Crystallography
Reprint in PDF format
 The structure of canavalin from Jack Bean
The structure of canavalin from Jack Bean