 Introduction
Introduction

PEAQ DSC is a differential scanning calorimeter (DSC), measuring the difference in the heat required to increase the temperature of a sample and its reference. This techinque is appliable for studies of phase transition, melting point and such transitions involve energy or heat capacity changes.
For more detail, please refer to MicroCal PEAQ-DSC website.
 Important Notification:
Important Notification:
* (2023-05-01) After replacing the liquid syringe tool, the instrument is ready for service.
* (2022-11-11) PEAQ-DSC Automatic system is down because of an error,
"There was an error during clean and the operation could not complete. Please check the autosampler configuration.
Can not move to target injector LC1 with current needle guidetype NotSpecified" Service is called. Sorry for the inconvenience.
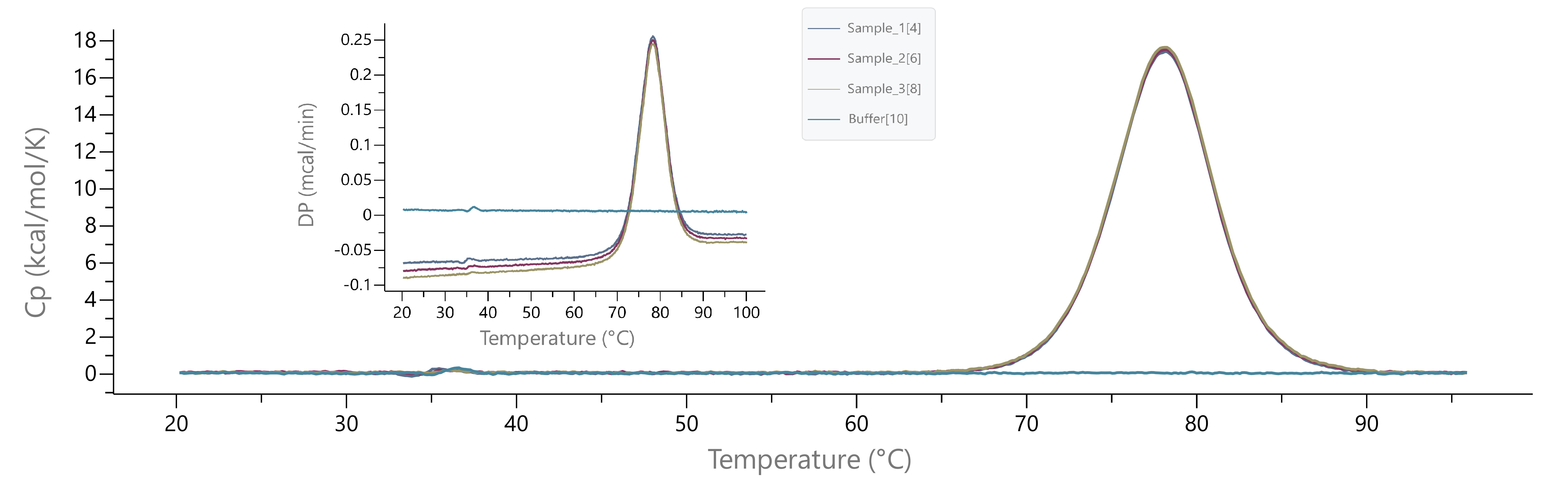
* (2022-02-21) Please check lysozyme scans below for recent instrument baseline before submitting samples to BCF. You may download the picture for better resolution.
* (2018-08-01) Automatic MicroCal PEAQ-DSC is available for sample submission.
* (2016-11-01) Free VP-Cap DSC sample tests available for Sinica users only.
 Features
Features
- Sample requirement ~ 325 μl of 0.1 - 1 mg/mL proteins.
- Non-reactive Tantalum 61™ capillary cells.
- Operating temperature range of 10ºC to 120ºC
(Ask BCF staff for temperatures outside this range). - Peltier element for precise temperature control.
- User selectable temperature scan rates, 0 to 240ºC per hour upscans.
- Pressurizing system (~60 psi).
 Applications
Applications
- Protein stability and folding.
- Liquid biopharmaceutical formulations.
- Process development.
- Protein engineering.
- Antibody domain studies.
- Characterization of membranes, lipids, nucleic acids and micellar systems.
- Assessment of the effects of structural change on a molecule’s stability.
- Assessment of biocomparability during manufacturing.
 Suggested Reading:
Suggested Reading:
VP-Cap DSC Microcalorimeter User's Manual.
DSC Data Analysis in Origin Tutorial Guide.
Cooper A, Nutley MA, and Wadood A 2001, Differential scanning microcalorimetry In Protein-Ligand Interactions: hydrodynamics and calorimetry, Harding SE, Chowdhry BZ (eds). Oxford University Press; pp.287-318.

